Posted on April 19, 2023 by Jared Mauskopf
Healthcare Mobile App or Web App: Which Is Better?
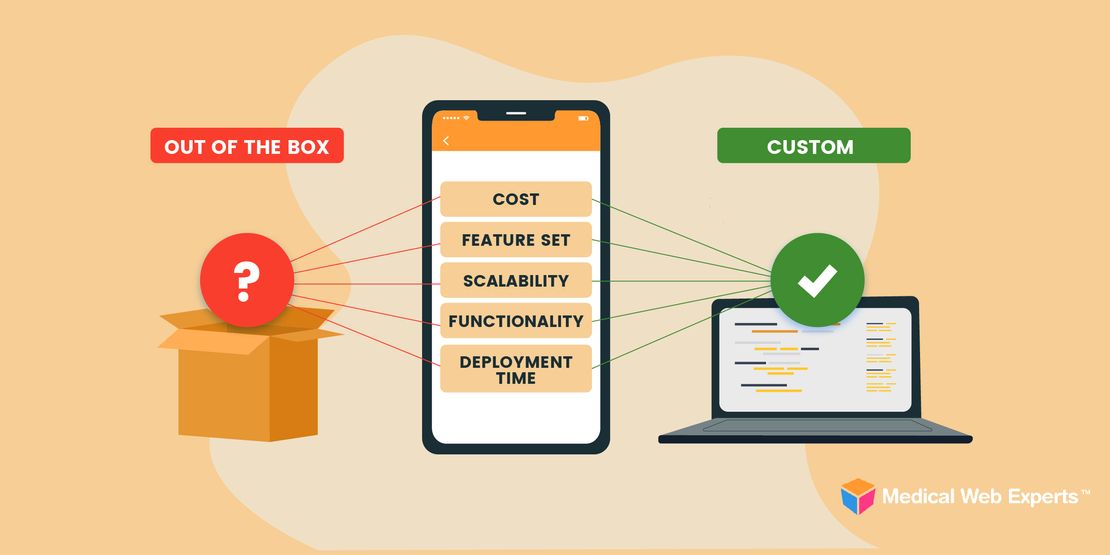
Healthcare organizations are increasingly under enormous pressure to offer the best digital tools to their patients. But before you set out to develop a healthcare application, you’ll need to make…Read more